(±)12-HETE-d8 (Synonyms: (±)12-Hydroxyeicosatetraenoic Acid-d8) |
| Catalog No.GC48738 |
Products are for research use only. Not for human use. We do not sell to patients.
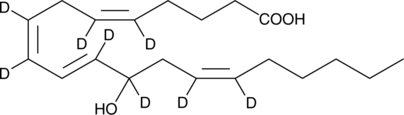
Cas No.: 2525175-25-9
Sample solution is provided at 25 µL, 10mM.
(±)12-HETE-d8 is intended for use as an internal standard for the quantification of 12-HETE by GC- or LC-MS. (±)12-HETE is formed via non-enzymatic oxidation of arachidonic acid .1,2 12(S)- and 12(R)-HETE are formed by 12(S)- and 12(R)-lipoxygenase-mediated oxidation of arachidonic acid, respectively.3,4 12(R)-HETE can also be formed by oxidation of arachidonic acid mediated by cytochrome P450s (CYP450s).5
1.Powell, W.S., and Rokach, J.Biosynthesis, biological effects, and receptors of hydroxyeicosatetraenoic acids (HETEs) and oxoeicosatetraenoic acids (oxo-ETEs) derived from arachidonic acidBiochim. Biophys. Acta1851(4)340-355(2014) 2.Derogis, P.B.M.C., Chaves-Filho, A.B., and Miyamoto, S.Characterization of hydroxy and hydroperoxy polyunsaturated fatty acids by mass spectrometryBioactive lipids in health and disease21-38(2019) 3.KÜhn, H., Banthiya, S., and van Leyen, K.Mammalian lipoxygenases and their biological relevanceBiochim. Biophys. Acta1851(4)308-330(2015) 4.BÜrger, F., Krieg, P., Marks, F., et al.Positional- and stereo-selectivity of fatty acid oxygenation catalysed by mouse (12S)-lipoxygenase isoenzymesBiochem J.348(Pt 2)329-335(2000) 5.Powell, W.S., and Rokach, J.Biosynthesis, biological effects, and receptors of hydroxyeicosatetraenoic acids (HETEs) and oxoeicosatetraenoic acids (oxo-ETEs) derived from arachidonic acidBiochim. Biophys. Acta1851(4)340-355(2015)
Average Rating: 5 (Based on Reviews and 14 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *



















