Miransertib (ARQ 092) HCl (Synonyms: Miransertib) |
| Catalog No.GC25638 |
Miransertib (ARQ 092) HCl is a novel, orally bioavailable and selective AKT pathway inhibitor exhibiting a manageable safety profile among patients with advanced solid tumors.
Products are for research use only. Not for human use. We do not sell to patients.
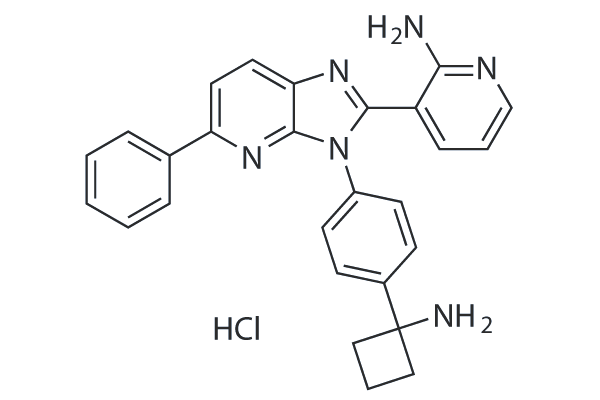
Cas No.: 1313883-00-9
Sample solution is provided at 25 µL, 10mM.
Miransertib (ARQ 092) HCl is a novel, orally bioavailable and selective AKT pathway inhibitor exhibiting a manageable safety profile among patients with advanced solid tumors.
ARQ 092 blocks membrane translocation of inactive AKT and even dephosphorylates the membrane-associated active form, thereby perturbing AKT activity. Treatment with 50-500 nM ARQ 092 significantly blocks αMβ2 integrin function in neutrophils and reduces P-selectin exposure and glycoprotein Ib/IX/V-mediated agglutination in platelets[2]. In a large panel of diverse cancer cell lines, ARQ 092 inhibits proliferation across multiple tumor types but are most potent in leukemia, breast, endometrial, and colorectal cancer cell lines. Moreover, inhibition by ARQ 092 is more prevalent in cancer cell lines containing PIK3CA/PIK3R1 mutations compared to those with wt-PIK3CA/PIK3R1 or PTEN mutations[1]. ARQ 092 targets the PI3K/AKT pathway and AKT specifically and reduces phosphorylation of GSK3α and GSK3β in mutation-positive cells[3].
Short-term oral administration of ARQ 092 or hydroxyurea, a main therapy for sickle cell disease, diminishes heterotypic cell-cell interactions in venules of sickle cell disease mice challenged with TNF-α. ARQ 092 is well tolerated at a continuous daily dose of 60 mg or a dose of 600 mg when administered once a week, for several months. ARQ 092 is likely to inhibit the activity of all AKT isoforms in intravascular cells and thereby attenuates the process of thrombosis and inflammation in SCD patients[2]. ARQ 092 is highly active in a subset of endometrial tumors that harbor PI3K pathway gene mutations[1].
[1] Yu Y, et al. PLoS One. 2015, 10(10):e0140479. [2] Kim K, et al. Haematologica. 2017, 102(2):246-259. [3] Lindhurst MJ, et al. Sci Rep. 2015, 5:17162.
Average Rating: 5 (Based on Reviews and 30 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















