Tropifexor (Synonyms: LJN452) |
| Catalog No.GC19363 |
Tropifexor (LJN452) est un agoniste très puissant du FXR avec une EC50 de 0,2 nM.
Products are for research use only. Not for human use. We do not sell to patients.
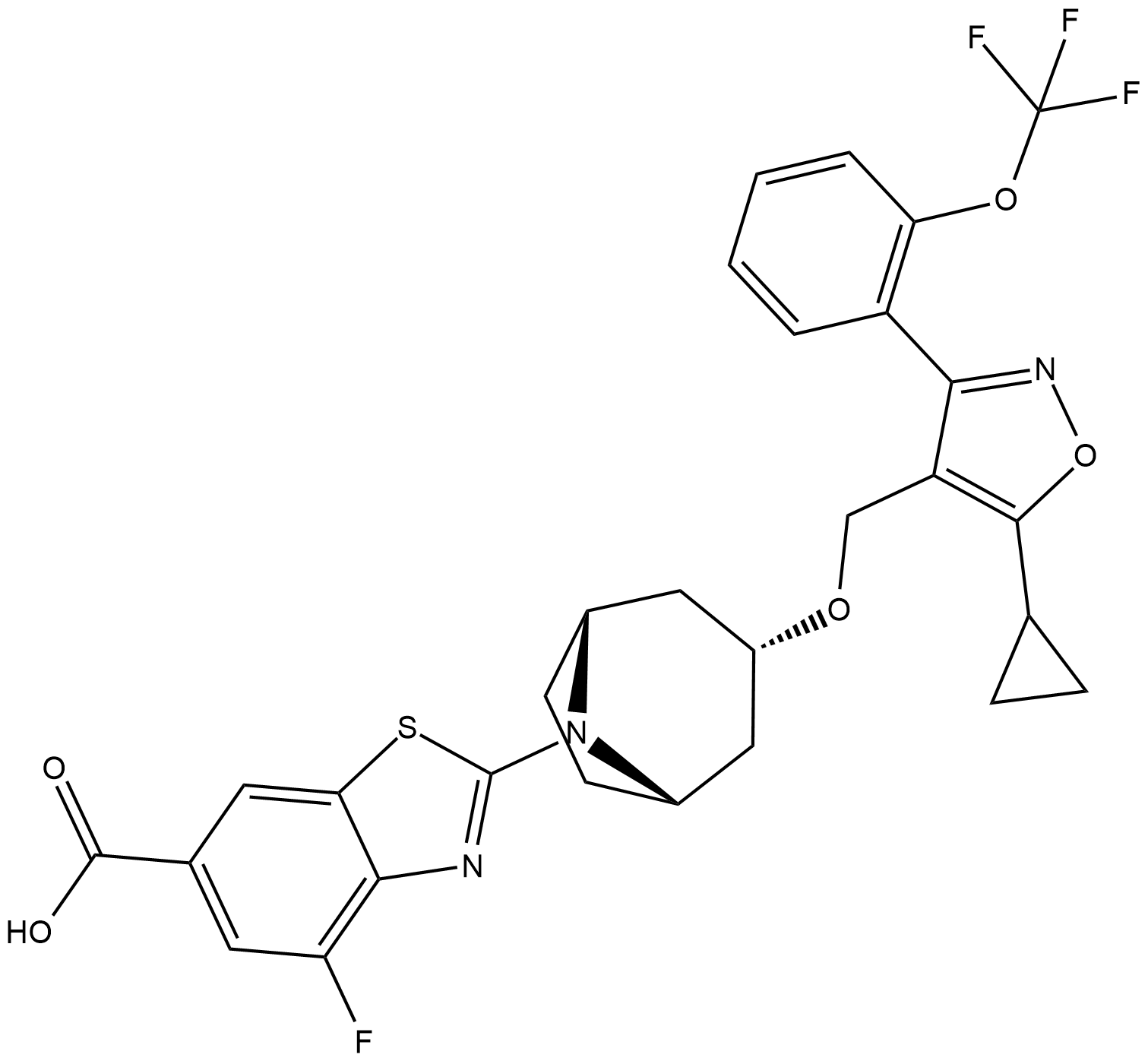
Cas No.: 1383816-29-2
Sample solution is provided at 25 µL, 10mM.
Tropifexor is a novel and highly potent agonist of FXR with an EC50 of 0.2 nM.
Tropifexor (compound 1) is a novel and highly potent agonist of FXR with an EC50 of 0.2 nM. Robust induction of both BSEP and SHP genes is observed in primary cells by Tropifexor in a concentration-dependent manner. BSEP induction above vehicle (DMSO) control is observed at concentrations as low as 1 nM, while strong induction of SHP (15-fold above vehicle) is observed at 10 nM and modest induction of SHP at 1 nM (3-fold)[1].
Tropifexor (compound 1) demonstrates highly potent induction of SHP and FGF15 in the ileum as doses as low as 0.1 mg/kg. In the liver, robust induction of SHP is observed at 0.01 mg/kg of Tropifexor with maximal levels of gene induction achieved at 0.3 mg/kg. Expression of CYP8B1 mRNA following 14 day treatment with Tropifexor is already apparent at the lowest dose (0.003 mg/kg), and CYP8B1 gene expression is fully repressed at doses above 0.03 mg/kg. Treatment of rats with Tropifexor exhibits a clear dose-dependent increase in plasma FGF15 protein, with maximal levels of FGF15 detected at 7 h postdose.Treatment with Tropifexor for 14 days produces a robust dose-dependent reduction in serum triglycerides and reaches a maximal response with a 0.3 mg/kg dose, resulting in a decrease of triglyceride levels to approximately 79% below the vehicle control group[1].
References:
[1]. Tully DC, et al. Discovery of Tropifexor (LJN452), a Highly Potent Non-bile Acid FXR Agonist for the Treatment of Cholestatic Liver Diseases and Nonalcoholic Steatohepatitis (NASH). J Med Chem. 2017 Dec 8.
Average Rating: 5 (Based on Reviews and 5 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















