Bicalutamide (Synonyms: ICI 176334, ZD 176334) |
| Catalog No.GC12271 |
비칼루타미드는 경구 활성 비스테로이드성 안드로겐 수용체(AR) 길항제입니다. 비칼루타미드는 전립선암 연구에 사용할 수 있습니다.
Products are for research use only. Not for human use. We do not sell to patients.
Cas No.: 90357-06-5
Sample solution is provided at 25 µL, 10mM.
Bicalutamide is an active non-steroidal androgen receptor antagonist with IC50 value of 160 nM.[1]
The androgen receptor (AR) is a type of nuclear receptor. It is activated by binding of either of dihydrotestosterone or the androgenic hormones testosterone. Then, it will translocate into the nucleus as DNA-binding transcription factor which regulates gene expression. Androgen is critical for the maintenance and development of the male sexual phenotype through related gene. The androgen receptor (AR) is also related to the core mechanism of castration-resistant prostate cancer.[1]
Bicalutamide inhibits growth in prostate cancer cells (VCaP cells) which overexpress androgen receptor by directly binding to AR then mediates androgen-mediated gene transcription with IC50 value of 160 nM. Bicalutamide directly binds to androgen receptor with Ki value of 12.5μM. In prostate cancer cells, bicalutamide impairs DNA binding and nuclear localization. Bicalutamide and MDV3100 significantly inhibited R1881-induced VP16-AR–mediated transcription in HepG2 cells with an IC50 value of 0.2 μM.[1] Bicalutamide has been an molecular template for designing selective androgen receptor antagonist used to the treatment of prostate cancer cells.[2] Bicalutamide also induces cell death by a different pathway which is independent of mitochondrial membrane potential changes and Bcl-2 action.[1]
Bicalutamide significantly inhibited AR then decrease the tumor growth in murine xenograft models, male immunodeficient mice which harbors LNCaP/AR-luc xenograft tumors.[1]
References:
[1]. Clegg NJ, Wongvipat J, Joseph JD, Tran C, Ouk S, Dilhas A, Chen Y, Grillot K, Bischoff ED, Cai L et al: ARN-509: a novel antiandrogen for prostate cancer treatment. Cancer Res 2012, 72(6):1494-1503.
[2]. Yin D, He Y, Perera MA, Hong SS, Marhefka C, Stourman N, Kirkovsky L, Miller DD, Dalton JT: Key structural features of nonsteroidal ligands for binding and activation of the androgen receptor. Mol Pharmacol 2003, 63(1):211-223.
| Cell experiment [1]: | |
|
Cell lines |
VCaP or Hep-G2 cell lines |
|
Preparation method |
The solubility of this compound in DMSO is >21.5 mg/ml. General tips for obtaining a higher concentration: Please warm the tube at 37℃ for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below -20℃ for several months. |
|
Reacting condition |
10-11 -10-4 M |
|
Applications |
Bicalutamide was found to be able to inhibit the growth in prostate cancer cells (VCaP cells) with overexpressed androgen receptor through directly binding to AR then mediating androgen-mediated gene transcription. Moreover, bicalutamide could impair the DNA binding and nuclear localization in prostate cancer cells. In HepG2 cells, bicalutamide could also significantly inhibit R1881-induced VP16-AR–mediated transcription with an IC50 value of 0.2 μM. |
| Animal experiment [1]: | |
|
Animal models |
Male immunodeficient mice harboring LNCaP/AR-luc xenograft tumors |
|
Dosage form |
10 mg/kg/day, oral |
|
Application |
In a clinically valid murine xenograft model of human CRPC, bicalutamide showed greater efficacy than MDV3100. Maximal therapeutic response in this model was achieved at 30 mg/kg/d of bicalutamide, whereas the same response required 100 mg/kg/d of MDV3100 and higher steady-state plasma concentrations. |
|
Other notes |
Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
|
References: [1] Clegg NJ, Wongvipat J, Joseph JD, Tran C, Ouk S, Dilhas A, Chen Y, Grillot K, Bischoff ED, Cai L et al: ARN-509: a novel antiandrogen for prostate cancer treatment. Cancer Res 2012, 72(6):1494-1503. |
|
| Cas No. | 90357-06-5 | SDF | |
| Synonyms | ICI 176334, ZD 176334 | ||
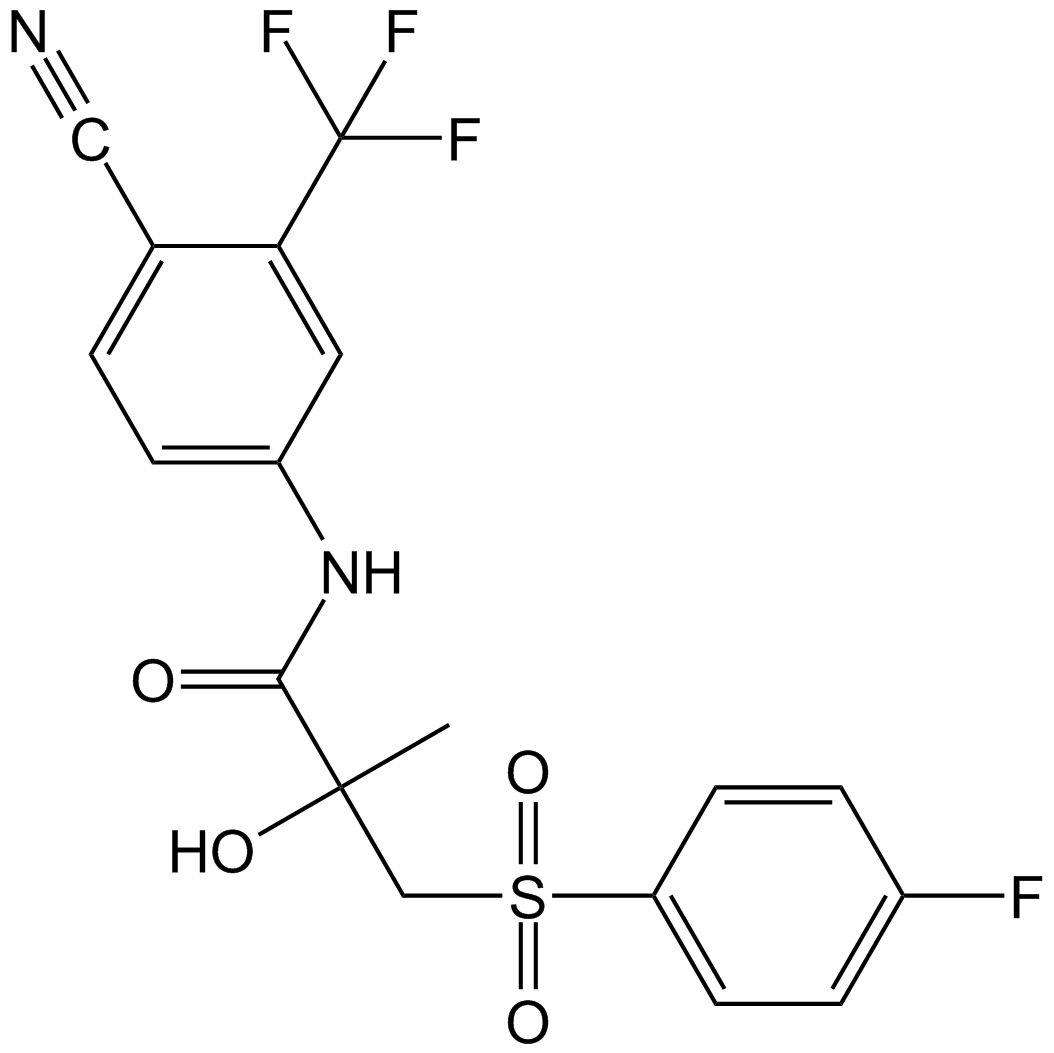
| Chemical Name | N-[4-cyano-3-(trifluoromethyl)phenyl]-3-(4-fluorophenyl)sulfonyl-2-hydroxy-2-methylpropanamide | ||
| Canonical SMILES | CC(CS(=O)(=O)C1=CC=C(C=C1)F)(C(=O)NC2=CC(=C(C=C2)C#N)C(F)(F)F)O | ||
| Formula | C18H14F4N2O4S | M.Wt | 430.37 |
| Solubility | ≥ 21.5mg/mL in DMSO, ≥ 4.3 mg/mL in EtOH with ultrasonic | Storage | Store at RT |
| General tips | Please select the appropriate solvent to prepare the stock solution according to the
solubility of the product in different solvents; once the solution is prepared, please store it in
separate packages to avoid product failure caused by repeated freezing and thawing.Storage method
and period of the stock solution: When stored at -80°C, please use it within 6 months; when stored
at -20°C, please use it within 1 month. To increase solubility, heat the tube to 37°C and then oscillate in an ultrasonic bath for some time. |
||
| Shipping Condition | Evaluation sample solution: shipped with blue ice. All other sizes available: with RT, or with Blue Ice upon request. | ||
| Prepare stock solution | |||

|
1 mg | 5 mg | 10 mg |
| 1 mM | 2.3236 mL | 11.6179 mL | 23.2358 mL |
| 5 mM | 0.4647 mL | 2.3236 mL | 4.6472 mL |
| 10 mM | 0.2324 mL | 1.1618 mL | 2.3236 mL |
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
 g
g
 μL
μL

Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such as vortex, ultrasound or hot water bath can be used to aid dissolving.
3. All of the above co-solvents are available for purchase on the GlpBio website.
Quality Control & SDS
- View current batch:
- Purity: >98.00%
- COA (Certificate Of Analysis)
- SDS (Safety Data Sheet)
- Datasheet
Average Rating: 5 (Based on Reviews and 6 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *



















