Application examples (from literature, for reference only)
Ref 1)
Yague P, Manteca A, Simon A, Diaz-Garcia ME, Sanchez J. New method for monitoring programmed cell death and differentiation in submerged S
treptomyces cultures. Appl Environ Microbiol.2010;76(10):3401-3404.doi:10.1128/AEM.00120-10
Experimental objective: To establish a simple and reliable method for monitoring and quantifying the cell death process of streptomycin fermentation products, using the live cell dye SYTO9 and propidium iodide PI. The principle of use is that SYTO9 is a nucleic acid dye with cell membrane permeability, which can label all cells: intact and damaged cell membranes. PI can only penetrate bacteria with incomplete membranes. Therefore, within the mixed bacterial community, bacteria with intact cell membranes exhibit green fluorescence, while bacteria with damaged membrane structures exhibit red fluorescence;
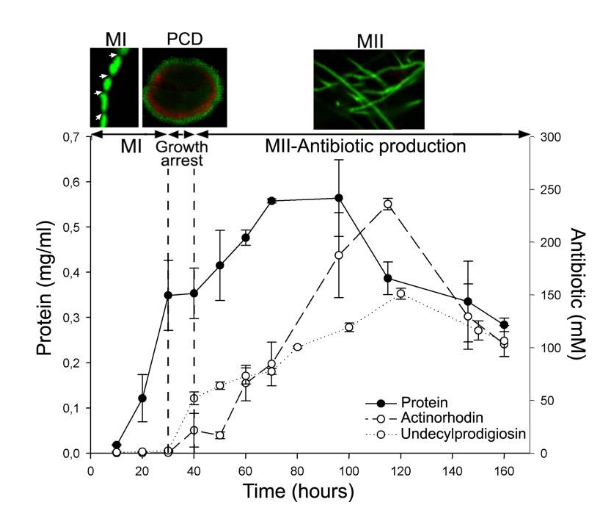
Fig. S. coelicolor growth curve and antibiotic (actinorhodin and undecylprodigiosin) production in submerged cultures. Confocal microscope images of key developmental stages stained with SYTO 9 and PI are shown at the top: individual hyphae of the first compartmentalized mycelium (MI; arrows indicate septation), second multinucleated mycelium hyphae (MII), and the mycelial pellet (240 μm in diameter) undergoing PCD processes in the center (red).
The transitory growth arrest phase coinciding with PCD is indicated. See text for details.
Ref 2 ) Robertson J,McGoverin C,Vanholsbeeck F and Swift S(2019) Optimisation of the Protocol for the LIVE/DEAD ® BacLightT™Bacterial Viability Kit for Rapid Determination of Bacterial Load. Front. Microbiol.10:801.doi: 10.3389/fmicb.2019.00801
Experimental method (SYTO 9+PI staining): Dilute SYTO 9 and PI with brown centrifuge tubes to concentrations of 33.4 and 400 μ M, respectively, and store on ice. Add 50 μ l of SYTO 9 and 50 μ l of PI working solution and/or 0.85% physiological saline (total volume of 100 μ l) to 0.9ml of culture, respectively, so that the concentrations of SYTO 9 and PI are 1.67 and 20 μ M, respectively. For the optimization experiment of the basic culture medium, each group of live and dead cell suspensions were stained with SYTO 9, PI, and SYTO 9+PI, respectively.
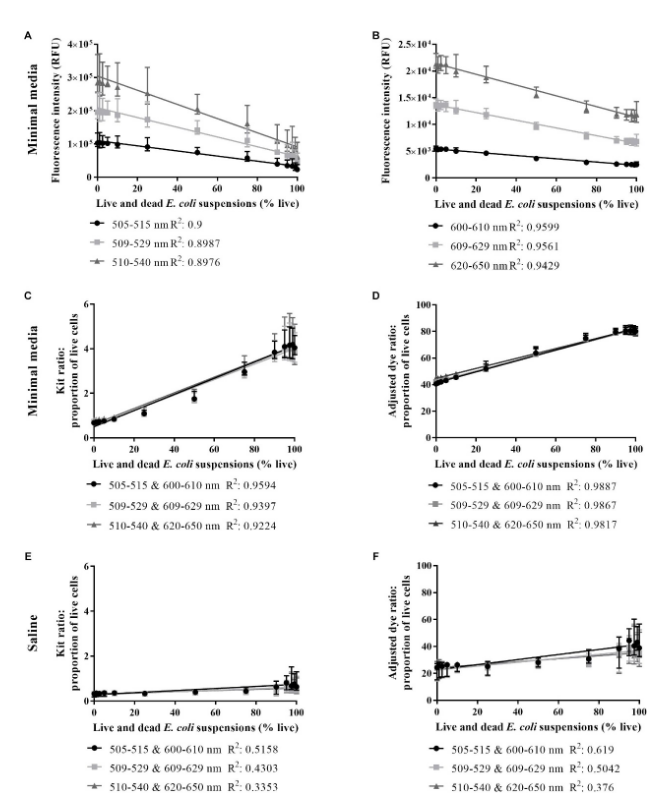
Fig. Live/dead spectrometry on live and dead E. coli suspensions in minimal media and saline.SYTO 9 intensity in minimal media(A), PI intensity in minimal media(B), proportion of live cells in minimal media derived from the kit ratio (C), % live cells in minimal media derived from the adjusted dye ratio (D), the proportion of live cells in saline derived from the kit ratio (E) and % live cells in saline derived from the adjusted dye ratio (F) for live and dead E. colisuspensions derived from integrating at three different wavelength ranges for each dye. An R2-value was generated from a linear regression ana
lysis of data from each wavelength range.Data presented is the median with the range from six biological replicates.
Dyeing process
Based on laboratory experience and published methods, it is recommended to use broad-spectrum staining concentrations for use, and optimize and explore the optimal working concentration according to one's own cell type and experimental system (see Table 2).
Dilute the SYTO 9 dye using a plastic tube, as the diluted dye will adhere to the glass. Overall, staining with phosphate free buffer can yield the best results. Residual cleaning agents on plastic or glassware are also present
It is possible to affect the true staining of many cells or organisms, resulting in bright fluorescent materials being visible in solutions containing or without cells. Ensure to clean the glassware with a mild detergent and rinse thoroughly with hot tap water,
Finally, clean several times with deionized water.
① Centrifuge cells and resuspend them with physiological salt solution or water. Adherent cells (such as mammalian cells) may be stained in situ on a cover glass. Use the recommended working concentration in Table 2 for staining. For the first experiment, it is recommended to
It is recommended to do multiple dye concentrations within the concentration range to determine the optimal working concentration for dyeing. Note that growth medium, cell density, presence of other cells, and other factors may all affect staining.
② Stained eukaryotic cells typically exhibit diffuse cytoplasmic and nuclear staining, especially with frequent bright intranuclear staining. Due to its cell membrane permeability and net positive charge at a neutral pH, this dye can also be Mitochondria can be stained, and mitochondria are mainly stained in live yeast.